Adsorbers
There are specific situations where engine operating conditions may not be ideal for conventional catalysts to achieve their full potential. Three-way catalysts, for instance, are highly effective in gasoline engine exhaust, but there is too much oxygen present in diesel exhaust for their NOx-reduction function to operate properly.
Also, catalysts generally need to reach a certain minimum temperature for effective operation, and although modern systems can reach this ‘light-off’ temperature within a few seconds there may still be some emissions until that temperature is reached.
Adsorbers offer ways to collect certain pollutants, specifically NOx or HC, during conditions which are not ideal, to temporarily store the pollutant and then to treat it when conditions are suitable. The two main current applications are NOx adsorbers, which can be used to treat NOx emissions from lean-burn gasoline engines or from diesel engines, and hydrocarbon adsorbers which can be used with conventional three-way catalysts to ‘trim’ the HC emissions from fuel enrichment needed for cold start.
NOx adsorbers
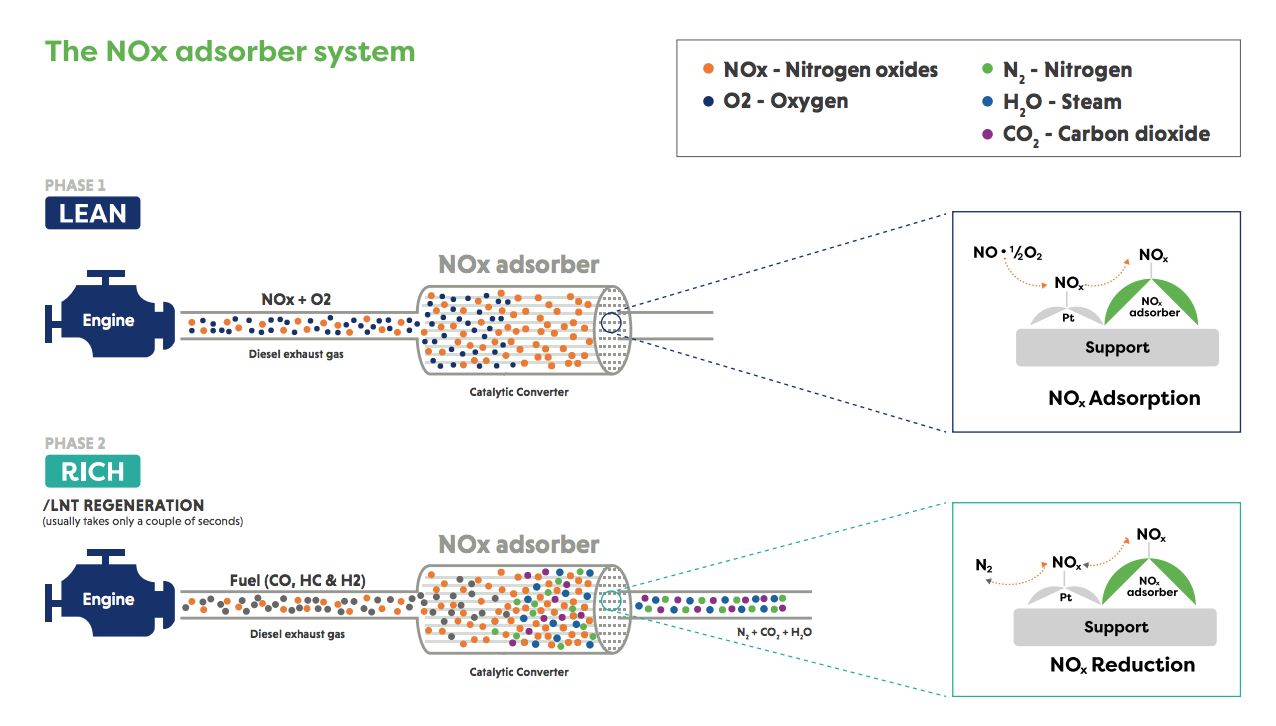
NOx adsorbers, also known as lean NOx traps (LNT) or NOx storage catalysts (NSC), adsorb and store NOx under lean conditions. A typical approach is to speed up the conversion of nitric oxide (NO) to nitrogen dioxide (NO2) using an oxidation catalyst so that the NO2 can be rapidly stored as nitrate on alkaline earth oxides. A brief return to stoichiometric* or rich operation for one or two seconds is enough to desorb (remove) the stored NOx and provide the conditions for a conventional three-way catalyst mounted downstream to reduce the NOx.
Stoichiometric conditions are those where the ratio of fuel and air is chemically correct for complete combustion. Under ‘lean’ conditions there is an excess of oxygen in the air:fuel ratio, under ‘rich’ conditions the opposite is true and there is more fuel than would be required for the chemically correct air:fuel ratio.
If the system settings are changed actively to switch between the lean and stoichiometric/rich conditions, it is labelled as an active NOx adsorber. Some applications make use of a passive NOx adsorber, which relies on a certain level of exhaust temperature to be reached to release the stored NOx.
Unfortunately, NOx adsorbers also adsorb sulfur oxides resulting from the fuel sulfur content. For that reason, fuels with a very low sulfur content (European ‘zero’ sulfur fuel contains less than 10 ppm sulfur) are required. The sulfur compounds are more difficult to desorb, so periodically the system has to automatically run a short ‘desulfation’ cycle to remove them.
Hydrocarbon adsorber systems
Hydrocarbon adsorber systems incorporate special materials, such as zeolites, into or upstream (in front) of the catalyst. Hydrocarbon emissions are collected when exhaust temperatures are too low for effective catalyst operation. The hydrocarbons are then desorbed at higher temperatures when the catalyst has reached its operating temperature and is ready to collect and oxidise the hydrocarbons.